The causes and consequences of fat malabsorption
Essential fatty acids are indispensable building blocks of human health
Fatty acids are derived from medium-chain triglycerides (MCTs) and long-chain triglycerides (LCTs). Omega-3 fatty acids benefit human health. LCTs are the ONLY source of omega-3 fatty acids.1
Omega-3 fatty acids strengthen the development and functioning of organs at the cellular level:1
![]()
Brain + Retina
Have highly specialized functional roles in normal signal transduction, neurotransmission, and neurogenesis2,3,4
![]()
Cardiovascular
Help lower blood pressure and improve blood vessel function5
![]()
Metabolic
Play a key role in numerous metabolic processes in addition to acting as a source of energy1,6
![]()
Systemic
Mediate immune function, platelet aggregation, and lipid homeostasis1,3
Omega-3 fatty acids can ease inflammation before it causes widespread damage to the body.2
- Docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) may have clinically important anti-inflammatory effects in a variety of conditions associated with fat malabsorption.2
- A balanced ratio of omega-6 to omega-3 fatty acids is beneficial to maintaining normal development, immunological function, and overall health.7,8
- Fats can increase caloric uptake and, when compared with proteins and carbohydrates, are a greater source of calories (9 kcal/gram vs 4 kcal/gram for proteins and carbohydrates).9
Fat malabsorption has devastating consequences
Gastrointestinal (GI) symptoms associated with fat malabsorption and EPI:10,11
- Diarrhea
- Steatorrhea
- Abdominal pain and discomfort
- Bloating and gas
- Weight loss
- Fatigue
Overall short- and long-term health impacts are multisystemic and varied:12,13
- Malnutrition
- Micronutrient deficiencies
(including fat-soluble vitamins) - Weight loss or inability to gain weight
- Pediatric developmental delays
- Death
Exocrine Pancreatic Insufficiency (EPI)
Fat malabsorption is associated with exocrine pancreatic insufficiency (EPI), a health condition where the body does not produce enough digestive enzymes or produces digestive enzymes that do not work properly. Children are most vulnerable to the consequences of untreated EPI and a delayed diagnosis often results in growth and developmental delays in addition to poor quality of life.14
Lipase is essential to break down fats into fatty acids and accounts for a majority of fatty acid digestion.15
- As pancreatic insufficiency progresses, lipid malabsorption causes many clinical symptoms and nutritional deficiencies while protein and starch digestion are usually maintained at a normal physiologic level.16
- Patients with compromised pancreatic output have a higher risk for fatty acid deficiencies, which may be related to a variety of adverse physiological effects, such as altered membrane and cellular functions.17
Conditions commonly associated with fat malabsorption and/or EPI:18–22
- Cystic fibrosis
- Acute/chronic pancreatitis
- Pancreatic and certain gastrointestinal cancers
- Trauma/critical care
- Short bowel syndrome
- Abdominal surgery
- Inflammatory bowel diseases
RELiZORB is formulated for enteral nutrition
In enteral formula, protein can be prepared in a form that is pre-hydrolyzed and available to be readily absorbed. However, pre-hydrolyzed fats in an absorbable form are not available in enteral formulas since they are not stable and spoil quickly.
RELiZORB connects directly to the feeding tube and is the only enzyme product designed to break down fats in enteral formula. The iLipase in RELiZORB is able to break down the triglycerides into an absorbable form (fatty acids and monoglycerides) before entering the body.
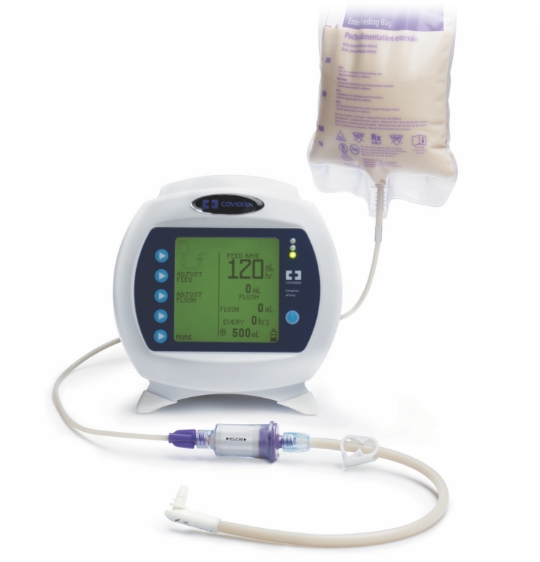
Pancreatic enzyme replacement therapy (PERT) capsules are not indicated for use with enteral formulas
PERT capsules are indicated for oral use only and are not intended to be crushed or added to enteral feeding formulas.
Manufacturer’s prescribing information advise against crushing PERT products and adding them to formula23
Crushing PERTs can result in overexposure of enzymes as well as clogged feeding tubes and accidental inhalation of crushed PERTs can be a risk to both patients and caregivers.24
There are no prospective clinical trials, efficacy data, or safety data evaluating or supporting the use of PERT with enteral nutrition.23,24,25
RELiZORB hydrolyzes fats prior to ingestion and is the only FDA-cleared enzyme product to hydrolyze fats in enteral formula.
RELiZORB is intended to provide continuous fat hydrolysis during enteral feeding

If oral PERT are taken during enteral feedings, frequent and consistent PERT dosing is needed for ongoing hydrolysis

RELiZORB is intended to provide continuous fat hydrolysis during enteral feeding
If oral PERT are taken during enteral feedings, frequent and consistent PERT dosing is needed for ongoing hydrolysis


*Timing is based on volume and flow rate. A single RELiZORB cartridge can be used for up to 500mL of formula at a rate of 10-120 mL/hr. A tandem RELiZORB cartridge configuration can be used for over 500mL and up to 1000mL of formula at a rate of 24-120 mL/hour. Please see additional details in the Instructions for Use.
References
- National Institutes of Health Website. Omega-3 fatty acids: fact sheet for professionals. https://ods.od.nih.gov/factsheets/omega3FattyAcids-HealthProfessional/. Accessed October 16, 2017.
- Calder PC. Omega-3 fatty acids and inflammatory processes. Nutrients. 2010;2(3):355-374.
- Abedi E, Sahari MA. Long-chain polyunsaturated fatty acid sources and evaluation of their nutritional and functional properties. Food Sci Nutr. 2014;2(5):443-463.
- Uauy R, Mena P, Rojas C. Essential fatty acids in early life: structural and functional role. Proc Nutr Soc. 2000;59(1):3-15.
- Harvard School of Public Health Website. The Nutrition Source. Omega-3 fatty acids: an essential contribution. https://www.hsph.harvard.edu/nutritionsource/omega-3-fats/. Accessed October 16, 2017.
- Prentice AM, Paul AA. Fat and energy needs of children in developing countries. Am J Clin Nutr. 2000;72(5 Suppl):1253S-1265S.
- Simopoulos AP. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed Pharmacother. 2002 Oct;56(8):365-79.
- Innis SM. Dietary omega 3 fatty acids and the developing brain. Brain Res. 2008 Oct 27;1237:35-43.
- Cleveland Clinic Website. Fat and calories. https://my.clevelandclinic.org/health/articles/fat-and-calories. Accessed October 16, 20.
- Blaauw R. Malabsorption: causes, consequences, diagnosis and treatment. S Afr J Clin Nutr. 2011;24(3):125-127.
- Alkaade S, Vareedayah AA. A Primer on exocrine pancreatic insufficiency, fat malabsorption, and fatty acid abnormalities. Am J Manag Care. 2017;23(supp 12):S203-S209.
- Dominguez-Munoz JE. Pancreatic exocrine insufficiency: diagnosis and treatment. J Gastroenterol Hepatol. 2011;26(2):12-16.
- Turck D, Braegger CP, Colombo C, Declercq D, Morton A, Pancheva R, Robberecht E, Stern M, Strandvik B, Wolfe S, Schneider SM, Wilschanski M. ESPEN-ESPGHAN-ECFS guidelines on nutrition care for infants, children, and adults with cystic fibrosis. Clinical Nutrition. 2016;35(3):557-577.
- Sankararaman S, Schindler T, and Sferra TJ. Management of Exocrine Pancreatic Insufficiency in Children. Nutrition in Clinical Practice. 2019;34(1):S27-S42.
- Whitcomb DC, Lowe ME. Human pancreatic digestive enzymes. Dig Dis Sci. 2007;52:1-17.
- Fieker A, Philpott J, Armand M. Enzyme replacement therapy for pancreatic insufficiency: present and future. Clin Exp Gastroenterol. 2011;4:55-73.
- Mogensen KM.Essential Fatty Acid Deficiency. Practical Gastroenterology. 2017;41(6):37-44.
- Singh VK, Haupt ME, Geller DE, Hall JA, Quintana Diez PM. Less common etiologies of exocrine pancreatic insufficiency. World J Gastroenterol. 2017; 23(39): 7059-7076.
- MedLinePlus Website. https://medlineplus.gov/ency/article/000299.htm. Accessed October 7, 2021.
- Capurso G., et al. Exocrine pancreatic insufficiency: prevelance, diagnosis, and management. Clin Exp Gastroenterol. 2019;12:129-139.
- Surmelioglu A, Ozkardesler E, Tilki M, Yekrek M. Exocrine pancreatic insufficiency in long-term follow-up after curative gastric resection with D2 lymphadenectomy: A cross-sectional study. Pancreatology. 2021 Aug;21(5):975-982.
- Blonk L, Wierdsma NJ, Jansma EP, Kazemier G, van der Peet DL, Straatman J, Exocrine pancreatic insufficiency after esophagectomy: a systematic review of literature. Diseases of the Esophagus. 2021 Dec;34(12).
- Schwarzenberg SJ. Enteral tube feeding for individuals with cystic fibrosis: Cystic Fibrosis Foundation Evidence-Informed Guidelines. Journal of Cystic Fibrosis 2016;15(6):724-735.
- Berry AJ. Pancreatic enzyme replacement therapy during pancreatic insufficiency. Nutr Clin Pract. 2014;29(3):312-321.
- Nguyen DL. Guidance for supplemental enteral nutrition across patient populations. Am J Manag Care. 2017 Jul;23(12 Suppl):S210-S219. PMID: 28727475.
- Cystic Fibrosis Foundation. https://www.cff.org/managing-cf/enzymes. Accessed April 7, 2023.